|
Since entropy changes are much smaller than enthalpy. The entropy change of a light rare-earth chloride on melting is therefore larger than that of a heavy rare-earth chloride. Hence, the magnitude of S for a reversible process such as a phase change is calculated. Any chemical or physical change in a system may be accompanied by either an increase in entropy (S > 0) or a decrease in entropy (S. When a system receives an amount of energy q at a constant temperature, T, the entropy increase S is defined by the following equation. Changes in entropy (S), together with changes in enthalpy (H), enable us to predict in which direction a chemical or physical change will occur spontaneously. Textbook content produced by OpenStax is licensed under a Creative Commons Attribution License. Entropy and the Second Law of Thermodynamics. Use the information below to generate a citation. Then you must include on every digital page view the following attribution: If you are redistributing all or part of this book in a digital format, Then you must include on every physical page the following attribution: 
If you are redistributing all or part of this book in a print format, Want to cite, share, or modify this book? This book uses the The net change in entropy of the system for the transition is The change in entropy for each step is Δ S i = Q i / T i. This can be accomplished experimentally by placing the system in thermal contact with a large number of heat reservoirs of varying temperatures T i T i, as illustrated in Figure 4.15. 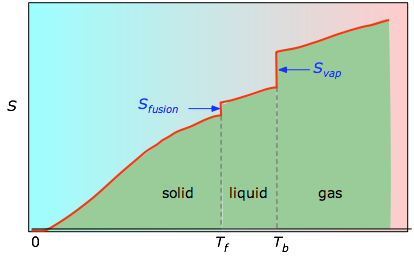
During each step of the transition, the system exchanges heat Δ Q i Δ Q i reversibly at a temperature T i. The temperatures associated with these states are T A T A and T B, T B, respectively. 
In order to account for spontaneity or directionality of processes, the concept of entropy is defined and incorporated into what is known as the second law of thermodynamics. S for isothermal expansion of an ideal gas. Is the process spontaneous at 10.00 C Is it spontaneous at +10.00 C Solution We can assess the spontaneity of the process by calculating the entropy change of the universe. Imagine a system making a transition from state A to B in small, discrete steps. Qualitative assessment of entropy changes. The entropy change for the process H 2 O ( s) H 2 O ( l) is 22.1 J/K and requires that the surroundings transfer 6.00 kJ of heat to the system. The change in entropy of a system for an arbitrary, reversible transition for which the temperature is not necessarily constant is defined by modifying Δ S = Q / T Δ S = Q / T. The same equation could also be used if we changed from a liquid to a gas phase, since the temperature does not change during that process either. Δ S = 16.8 kJ 273 K = 61.5 J/K Δ S = 16.8 kJ 273 K = 61.5 J/Kĭuring a phase change, the temperature is constant, allowing us to use Equation 4.8 to solve this problem.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
